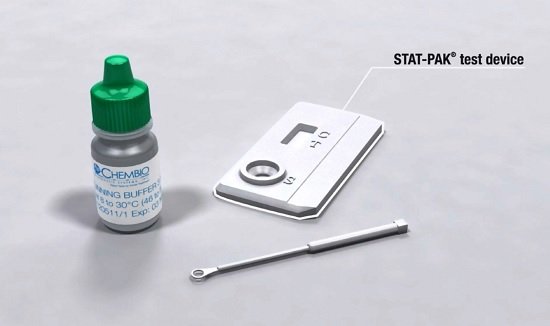
Chembio Diagnostics has obtained a key approval to manufacture HIV tests in Malaysia for the international market.
The Medford-based company said the World Health Organization approved its Malaysia factory through its prequalification program to produce Stat-Pak HIV 1/2 tests.
The company said this review included inspecting the manufacturing site, reviewing products, performance evaluation and labelling review.
“Our investment in Malaysia is a key element of our growth strategy, commercially and operationally, and the WHO prequalification approval represents a major milestone,” Chembio CEO John Sperzel said. “This approval allows the company to manufacture our Stat-Pak HIV 1/2 test in Malaysia and supply it to international markets.”
He said the company plans to do this during the fourth quarter of 2019 and to manufacture additional products at that factory.
Chembio in January 2017 acquired RVR Diagnostics, which had received funding from the Malaysian Technology Development Corporation and the Malaysian Investment Development Authority.
“Malaysia has a well-developed infrastructure, high-quality workforce and pro-business government policies, all of which offer significant strategic benefits for Chembio’s operations in the region,” according to the company.